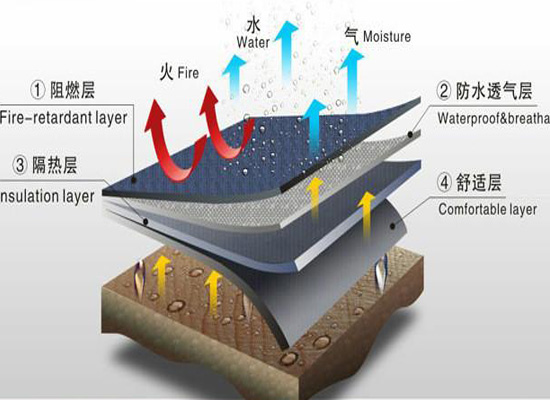
Do you really understand flame retardant fabrics?
Introduction:
With the development needs of the contemporary textile industry and people’s safety requirements, the role of flame retardants in fabrics has become indispensable. The most widely used ones are in the field of textiles. The most important prerequisite for flame retardant fabrics is the baking time and temperature, but the baking time and temperature depend on the structure of the fabric and the temperature of the pre-treatment.

Functional principles of flame retardants:
(1) Endothermic mechanism
Some flame retardants have an endothermic function. When they decompose when heated, they will absorb heat (aluminum hydroxide, magnesium hydroxide, etc.) and can cool the flame-retardant substrate to the temperature necessary to maintain combustion. below temperature. In addition, this type of flame retardant can also produce water vapor or other gases during the endothermic decomposition process, which isolates the substrate from the air and inhibits the further development of the combustion process.
(2)Shielding mechanism
The shielding mechanism is also called the isolation effect. This type of flame retardant is generally used in coatings and is one of the important components of coatings. When wood is heated or burned, the flame retardant component in the coating will release fire extinguishing gas to prevent it from burning. The coating expands to form a uniform and dense foam insulation layer, which isolates oxygen, inhibits the transfer of heat from the heat source to the substrate, delays the time for the flammable substrate to reach the ignition temperature, and prevents the spread of flames.
(3)Dilution mechanism
Flame retardants decompose under high combustion temperatures to produce a large amount of non-flammable gases, such as water, carbon dioxide, ammonia, etc. These non-combustible gases dilute the concentration of combustible gases in the gas phase, thereby inhibiting or terminating combustion.
(4) Inhibition mechanism
Polymer is an important component of this kind of flame retardant. The combustion of polymer is mainly a chain reaction produced by OH radicals. Such substances have the ability to react repeatedly with OH radicals to generate water, which can inhibit the generation of free radicals. chain reaction. For example, halogen flame retardants produce highly active free radical scavengers during combustion, which can capture flammable groups in the gas phase; phosphorus-containing flame retardants generate metaphosphoric acid during combustion, forming an isolation barrier to prevent building materials from burning. Exposed to the atmosphere to achieve flame retardant effect.
The heat setting and baking temperature and time of the flame retardant must be compatible with the structure of the fabric fiber. However, it is worth noting that the baking temperature of the flame retardant must be higher than the heat setting temperature of the fabric. Secondly, attention should be paid to After flame retardant treatment, flame retardant may remain on the surface of the fabric, so be sure to clean it thoroughly!

The best prerequisites for flame retardant finishing are:
The fabric must be clean and absorbent to ensure that the flame retardant can penetrate well and avoid possible chemical reactions between chemicals, such as sizing. These are all factors that affect the flame retardancy of fabrics. There may also be residual starch, slurry or oil after pre-treatment of fabrics, which must be washed clean.
Drying efficiency is usually higher than production efficiency. For production and processing, adding a large amount of finishing agents cannot make up for the lower processing effect. Production also needs to improve finishing stability and delay processing time.
Flame retardant finishing is a wet treatment process, and the pH value of the treatment solution needs to be adjusted to a suitable size before use.